Here we describe the synthesis and characterization of the activity based fluorescent and cell permeable inhibitor bodipy tmr ahx 3 l 3 vs mv151 which specifically targets all active subunits of the proteasome and immunoproteasome in living cells allowing for rapid and sensitive in gel.
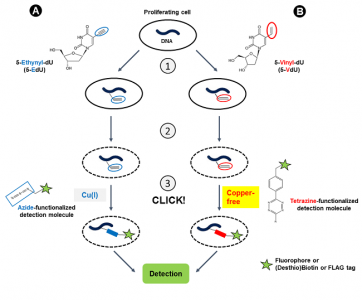
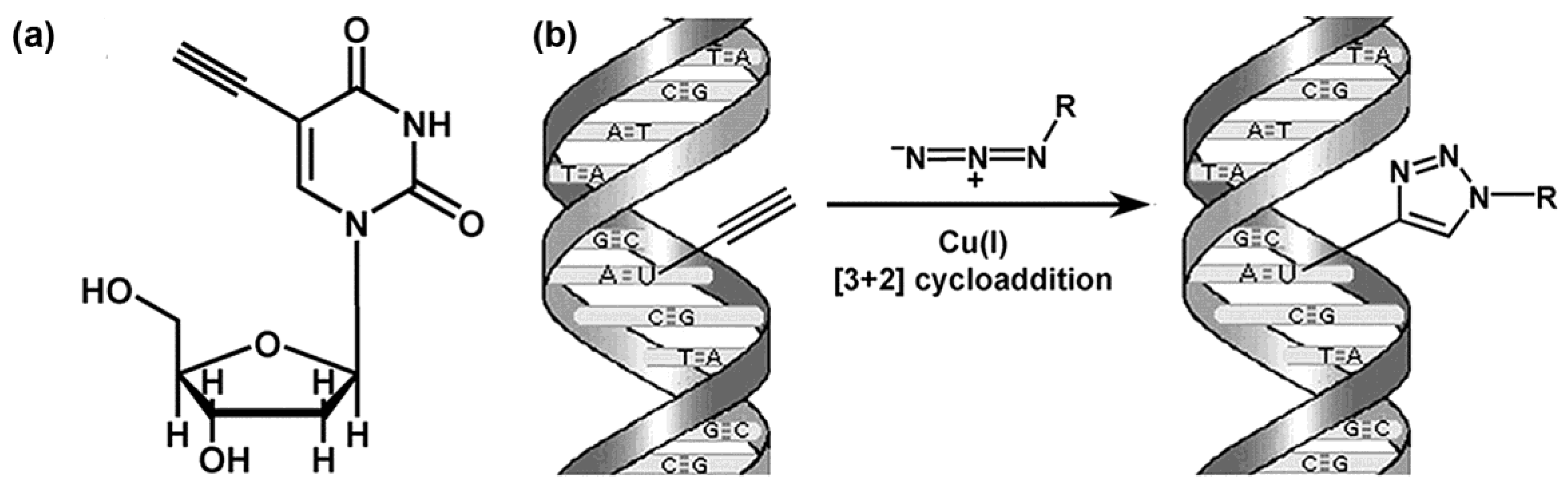
In vitro vinyl du labeling.
They repel everything from water to oil and chemicals.
Heavy duty white vinyl labels for laser printers.
Instrument diagnostique clinique in vitro medical devices regulations label.
Prepare a 10 mm stock solution of brdu ab142567 by dissolving 3 mg of brdu in 1 ml water.
A medical device or a product subject to section 3 1 of the medical devices regulations that is intended to be used in vitro for the examination of health canada labelling of in vitro diagnostics devices ivdds.
The proteasome is an essential evolutionary conserved protease involved in many regulatory systems.
Means an in vitro diagnostic device that is intended for use outside a laboratory for testing at home or at the point of care such as a pharmacy a health care professional s office or the bedside.
In vitro diagnostic ivd labeling requirements are located in 21 cfr part 809.
Numbers appearing in parentheses next to subject headings are the corresponding sections of 21 cfr.
Random labeled single stranded rna probes can be synthesized by in vitro transcription.
Near patient in vitro diagnostic device near patient ivdd.
Incubation time may vary from a few minutes to 20 minutes depending on the cell types.
The reaction is catalyzed by bacteriophage t7 rna polymerase that incorporates labeled ntps mostly utp as substitute for their natural counterpart using linear rna probe encoding dna as template.
These bright white labels with a matte finish are designed for rugged applications where a waterproof label is required.
Dilute the 10 mm brdu stock solution in cell culture medium to make a 10 µm brdu labeling solution.
In vitro diagnostic device ivdd.
The optimal ratio of labeled ntp ntp in terms of product yield and labeling efficiency depends on both the.
In vitro labeling of cells with brdu.
Filter the 10 µm brdu labeling solution through a 0 2 µm filter under sterile conditions.
Food and drug administration 10903 new hampshire avenue silver spring md 20993 1 888 info fda 1 888 463 6332 contact fda.
Convey some of the information required for in vitro diagnostic devices ivds intended for professional use by 21 cfr 809 10 fda s labeling requirements for in vitro diagnostic devices and 21 cfr parts 610 and 660 fda s labeling requirements for biologics including ivds that are licensed under the public health service phs act.